ECCS B320
The ECCS B320 is an electro-chemical reaction visualizing confocal system that enables operand observation of lithium-ion batteries. It observes the electrode cross-section in real-time during charge/discharge cycles and visualizes the internal reactions of the battery.
With the real-color confocal optical system, it can observe color changes in electrode materials with high precision, providing important information for battery performance evaluation and research and development.

Product Overview
Operand observation of electrode cross-section during charge/discharge
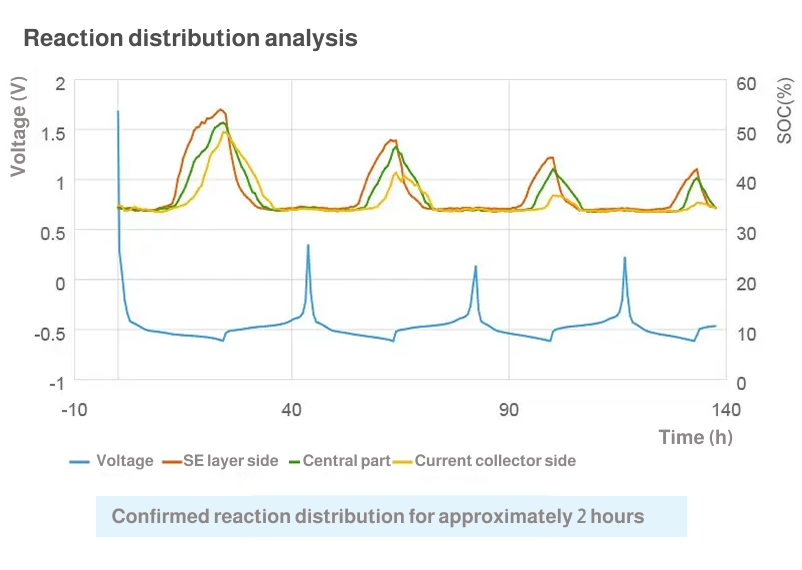
Analysis of reaction distribution in thickness direction for each electrode
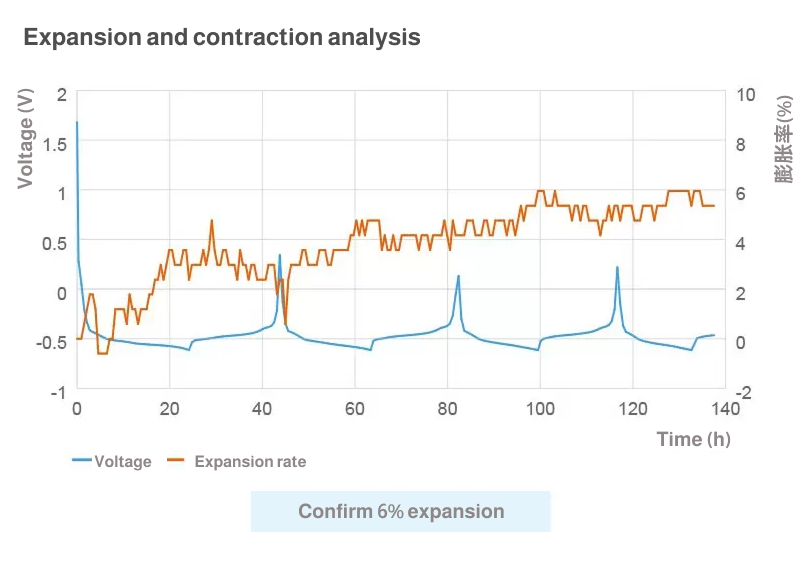
Measurement of electrode expansion and contraction
Visualization of dendrite formation process
Applications
| Overall battery performance |
Visualization of dendrite formation mechanism Analysis of electrochemical reactions at different C-rates Analysis of capacity degradation from cycling Analysis of temperature characteristics Evaluation of next-generation batteries including sodium-ion battery, solid state battery, Li metal anodes, and metal-air battery Applicable to both full cell and half cell |
| Anode |
Visualization of intercalation Analysis of the color of graphite anode in various states of charge/discharge Measurement of expansion and contraction amount Visualization of dendrite formation mechanism |
| Cathode |
Analysis of lithium-ion absorption and desorption Measurement of expansion and contraction amount |
| Electrolyte |
Visualization of gases being generated and identification of their sources Evaluation of the effect of additives |
| Conductive additives |
Indirect evaluation of the dispersed state from changes in electrode color and brightness Evaluation of the effect of carbon nanotubes |
| Binder |
Verification of binding between active materials Verification of binding between collectors and active materials Evaluation of cycle characteristics by binding property |
| Separator |
Verification of the effect of chosen separator materials on reaction distribution Verification of the effect of ceramic and other coatings on reaction distributions |
Options
Real-color confocal optics
Real-color confocal optics is designed to collect reflected light from the focal plane (the surface of active materials on electrode) without being affected by the glass surface or electrolyte, providing high-resolution color images with good color separation.
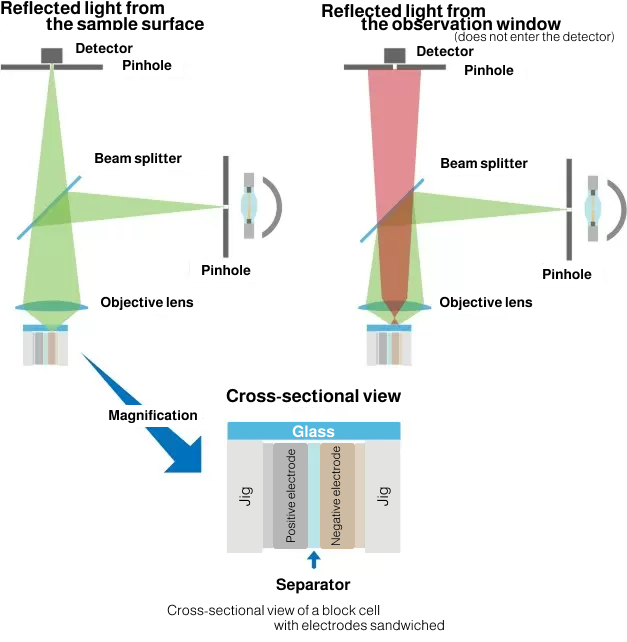
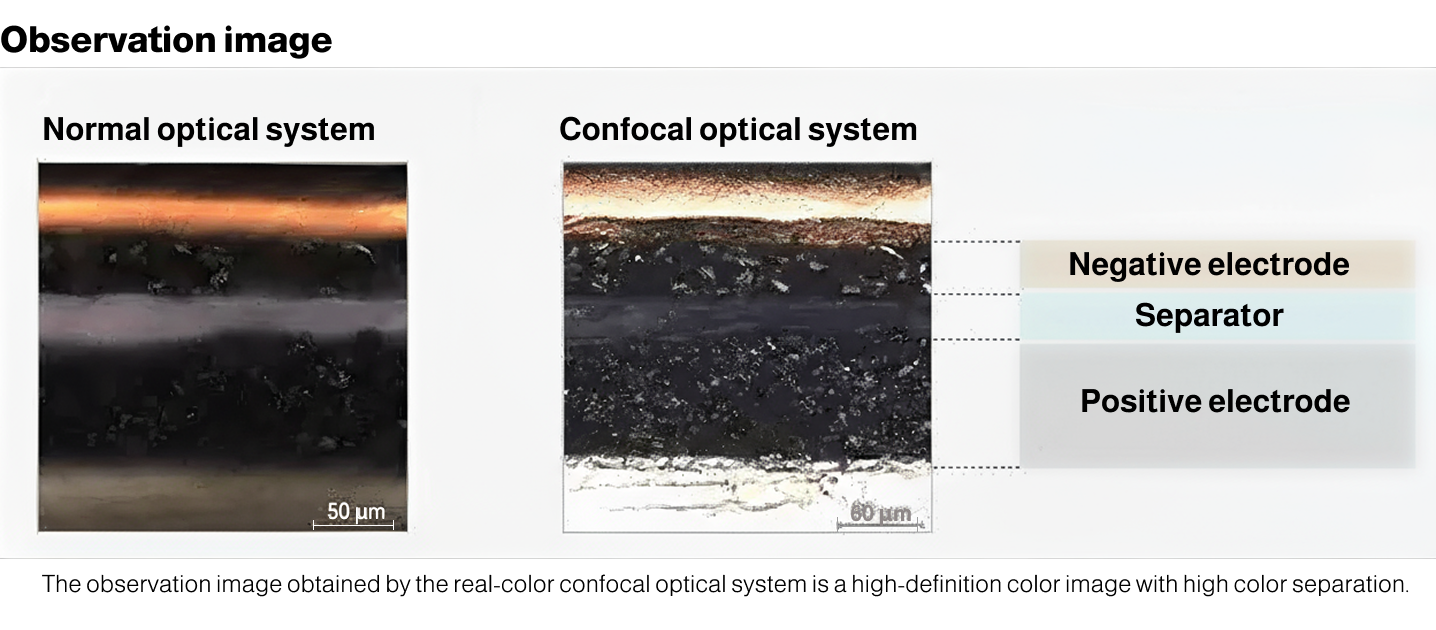
The observation images provided by real color confocal optics are high-definition color images with good color separation
Observation cell
The following specially-designed observation cells are available for in-situ observation:
Observation cell for coin cell
Basic model compatible with coin-shaped electrodes
Observation cell for pouch cell
Advanced model compatible with stacked batteries, etc.

Temperature control system
Specially-designed thermostatic tank that can store the observation cell
The inside of thermostatic tank is kept dry to prevent condensation.
Range of temperature control: -30℃~80℃

Analysis of electrochemical reaction distribution
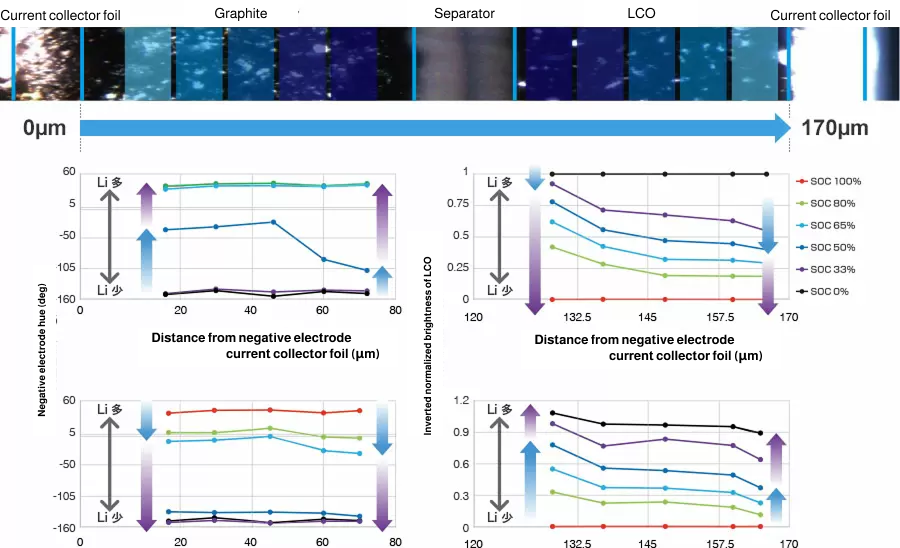
The graphite anode goes through several different stages during charge and reaches SOC100% when the intercalation of lithium ions occurs between all layers. In the process, the color of active materials changes from grey to blue, red and finally gold. From these colors, you can analyze the electrochemical reaction distribution quantitatively.

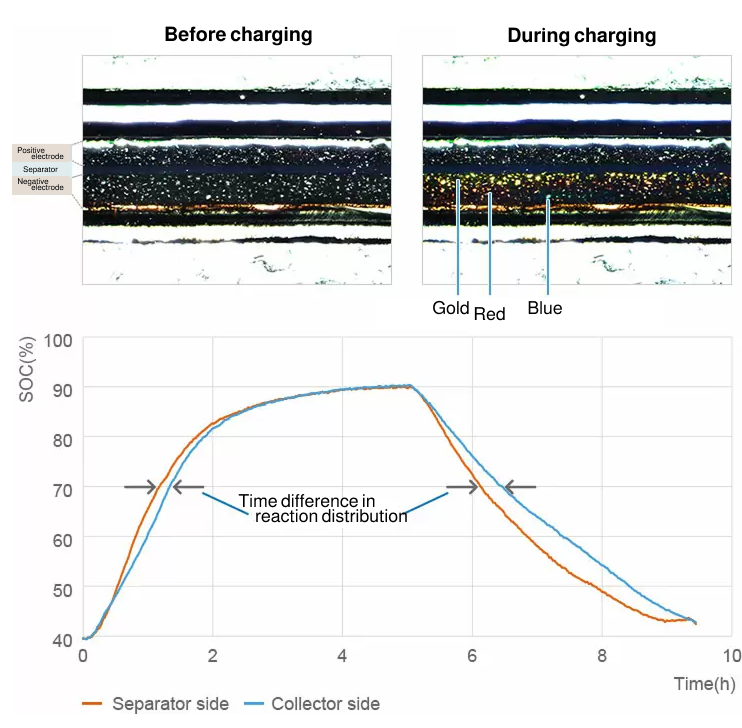
The change of color seen in graphite anodes will not usually occur in cathodes, but you can quantitatively analyze the reaction distribution in cathodes by measuring small changes in brightness during charge/discharge.
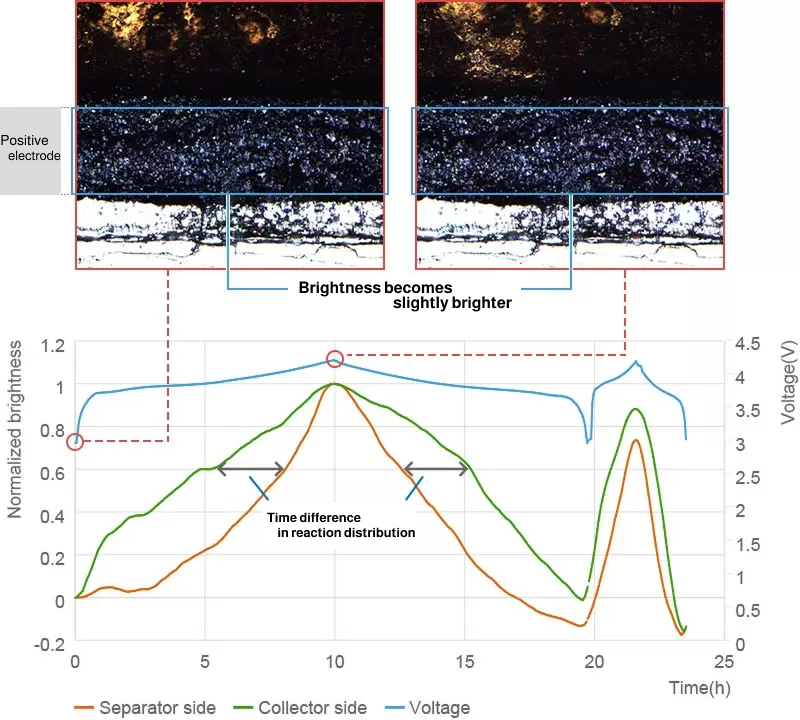
From presentation at Battery Symposium in Japan 2017 (2A14)
The analysis of reaction distribution for both cathode and anode is possible at the same time-the former based on change in brightness and the latter based on change in hue.
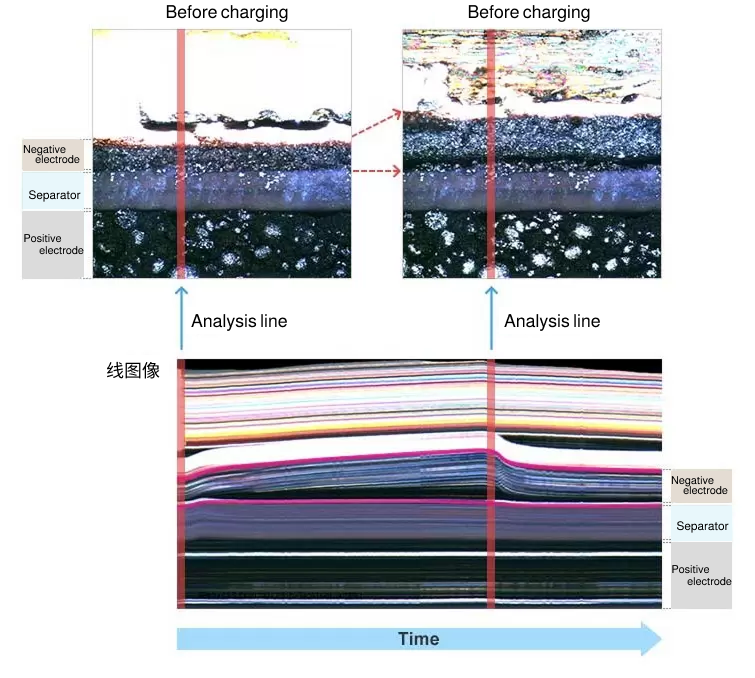
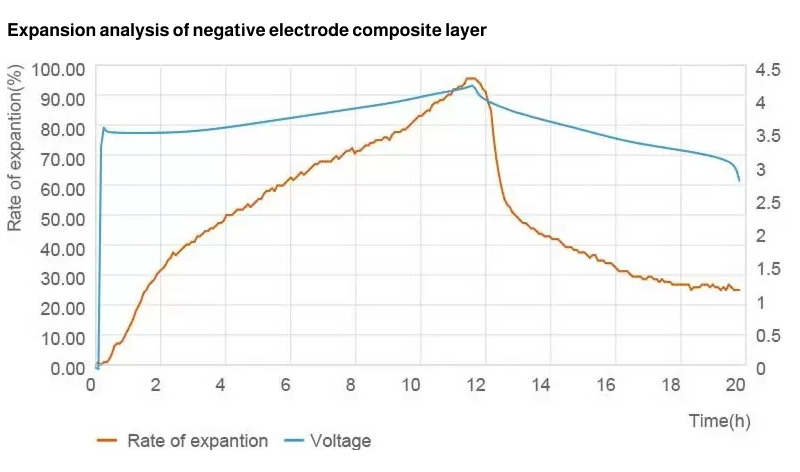
Analysis of expansions and contractions
The expansion/contraction analysis using ECCS enables small-change measurement and real-time analysis, neither of which is possible with a micrometer or displacement gauge. ECCS can create a "line image" by slicing a captured image at a user-specified line of analysis and by aligning successive sliced images in a chronological order. You can select a mixture layer and conduct measurement on the line image.
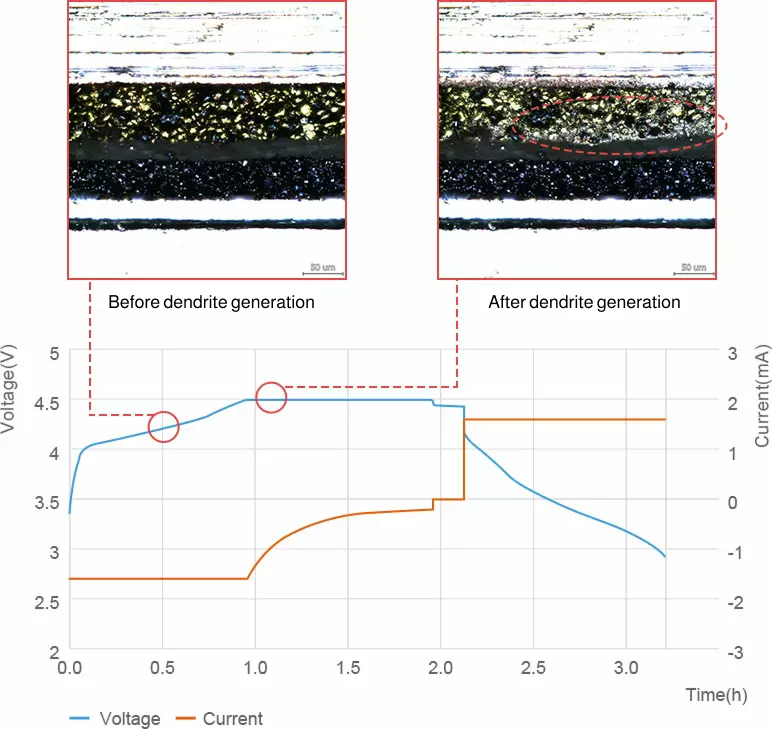
Observation of dendrite formation
Evaluation of dendrite formation mechanism
ECCS enables the observation of small dendrite formations that are not visible in the charge/discharge curves of batteries.
Measurement of voidage in electrode
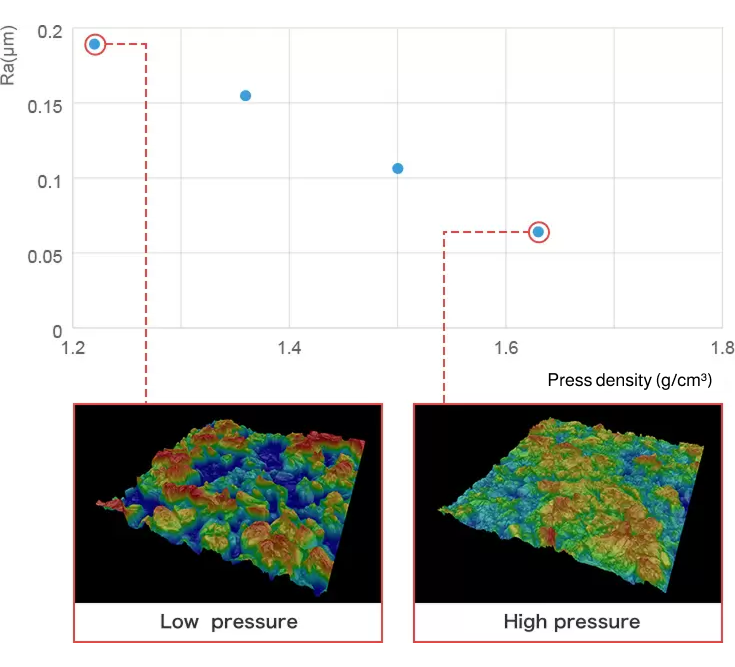
ECCS B320 has a microscope function that can be used to measure the surface roughness and voidage of electrode.
Solid state battery
Sample by Prof. Hayashi, Osaka Prefecture University
※Click the screen to watch the video.
※This is a fast-forwarded replay.
| Working electrode | Graphite :SE = 50:50(wt.%) |
| Counter electrode | Li-In |
| Solid state electrolyte | Li2S・P2S5 |
| C-rate | 0.03C |
Stacked battery
The newly released laminated cell can be used for analysis of double-side-coated electrodes and Stacked battery.
※Click the screen to watch the video.
※This is a fast-forwarded replay.
Graphite
High magnification
ECCS B320 provides high-magnification color images and supports the analysis of electrochemical reactions of an active material.
| Working electrode | Spheroidal graphite |
| Counter electrode | Li箔 |
| Charge rate | 0.3C |
| Cell | Block cell for cross-sectional observation |
Reaction distributions
ECCS B320 shows dendrites being formed by current concentration as a result of uneven reactions during the charging process.
※Click the screen to watch the video.
※This is a fast-forwarded replay.
| Anode | Artificial graphite |
| Cathode | NCM |
Silicon anode
ECCS B320 visualizes the volumetric expansion of active materials and enables you to measure the thickness of active materials for analysis and comparison with the battery's charge/discharge curves.
※Click the screen to watch the video.
※This is a fast-forwarded replay.
| Anode | Si |
| Cathode | NCA |
Dendrite
ECCS B320 shows dendrites being formed while a battery is overcharged at high rate. Once the charging stops, the dendrites start to shrink and lithium ions are dispersed from dendrites to charge the anode.
| Anode | Spheroidal graphite |
| 正極 | Cathode |
| Charge rate | 4C |
| Cell | Block cell for cross-sectional observation |
※Click the screen to watch the video.
※This is a fast-forwarded replay.
LCO
ECCS can capture small changes in brightness during charge/discharge and enables you to make quantitative analysis of the reaction distribution in the cross section of cathodes in the same way you do for anodes.
※Click the screen to watch the video.
※This is a fast-forwarded replay.
NMC
ECCS can capture very small expansions and contractions in cathodes, which usually show only a slight dynamic change.
※Click the screen to watch the video.
※This is a fast-forwarded replay.
Outgassing
ECCS enables the observation of outgassing during charge/discharge. In-situ gas analysis is also available when a gas analysis cell is used (together with a gas chromatograph).
※Click the screen to watch the video.
※This is a fast-forwarded replay.
